About
We were built around a simple premise:
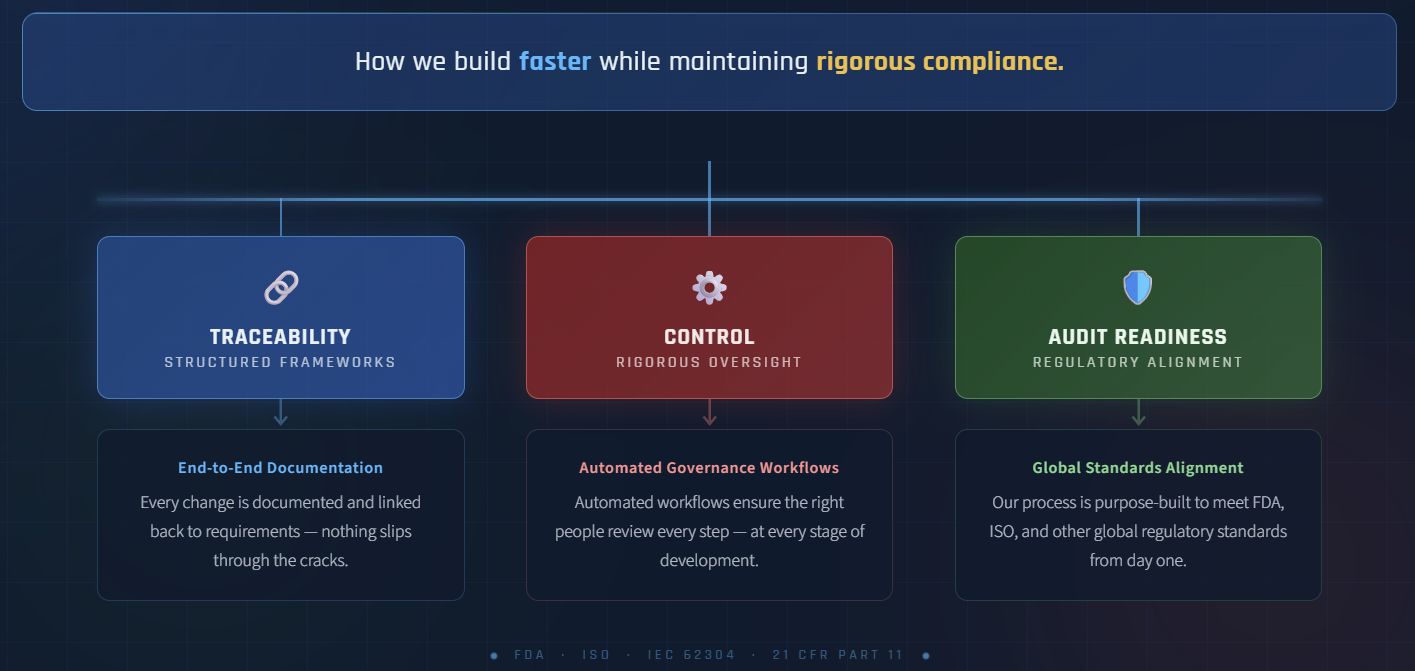
Move faster, compliantly — without sacrificing traceability, control, or audit readiness.

Compliance should not slow innovation.
It should strengthen it.
We integrate regulatory architecture directly into engineering execution — aligning ISO 13485 quality systems, IEC 62304 lifecycle controls, ISO 14971 risk management, and structured verification practices from the start.
The result is disciplined acceleration.
Not shortcuts.
Not document inflation.
Not last-minute remediation.
Structured systems that are:
Traceable from requirement to validation
Risk-aligned by design
Audit-ready at every lifecycle stage
Defensible under regulatory scrutiny
Combining regulatory systems thinking with hands-on validation engineering.
We do not separate documentation from implementation.
We do not treat QMS as a static binder.
We do not bolt compliance onto finished software.
Instead, we build integrated frameworks that connect:
Lifecycle planning
Risk-based decision making
Design controls
Verification & validation
Cybersecurity governance
Post-market monitoring
All within a structured, scalable system.
Who We Serve.
AI is part of our methodology — but never a substitute for discipline.
We use AI to:
Accelerate structured documentation
Reinforce traceability mapping
Strengthen risk identification
Improve testing coverage analysis
But every output is governed by controlled processes, review discipline, and regulatory alignment.
Speed matters.
Control matters more.
SaMD startups preparing for first submission
Scaling digital health platforms strengthening QMS maturity
AI-enabled medical device teams requiring validation rigor
Established organizations optimizing lifecycle integration
Whether through full QMS systems, structured DIY frameworks, or hands-on advisory, our focus remains the same: Engineer compliance with clarity, precision, and defensibility.